A hydrogen atom is partially positively charged which therefore means that it is attracted to atoms which have a partial negative charge. These bonds involve the hydrogen atom and occur between hydrogen and other atoms. Salts are typically formed by ionic bonds being formed between atoms that are strongly different in charge for example, a positive sodium ion being attracted to a negative chloride ion to form the salt, sodium chloride. Ionic bonds are typically seen when there are even larger differences in electronegativity than is seen in polar covalent cases. Slight charge differences and differences in electronegativity result in a polar covalent bond being formed between the atoms in the reaction. If the electronegativity is similar then the sharing is said to be more equal and a nonpolar covalent bond is formed. The type of bond does vary depending on how different the electronegativity of the participating atoms are. The covalent bonds are common in many biological molecules, for instance, the glycosidic bonds formed between sugar molecules and the peptide bonds formed between amino acids. Ionic bonds occur when atoms have different charges and where there is a big difference in electronegativity between the two.Ĭovalent bonds form when atoms have similar electronegativities and involve a sharing of the valence electrons. There are three main types of bonds that are found in biological molecules, ionic, covalent and hydrogen bonds. Atoms need to fill the outermost or valence shell with electrons to achieve stability. There is variation in the types and strength of different chemical bonds that form between atoms.īonds often tend to form when atoms are unstable or where there are opposite charges on different atoms. Chemical bondsĪtoms join together by various types of bonds that form. Water is essential to life and enables organisms to break down large molecules into monomers by hydrolysis. These bonds are responsible for many of the properties of water such as cohesion and adhesion, and the ability of water to be a solvent for many other types of molecules. This means that the hydrogen of one molecule is attracted to the oxygen of another. The oxygen atom of water is partially negative while the hydrogen atoms are partially positive. Hydrogen bonds are how one water molecule is attracted to and attached to other water molecules. This is important in the case of nucleic acids such as DNA in which strands have to separate to allow for the processes of DNA replication and transcription. These bonds are weak and can break relatively easily as a result. Hydrogen bonds always include a hydrogen atom that is attached to one or more other atoms to form a molecule. Ionic bonds form when there are big differences in the electronegativity and the charges of atoms.

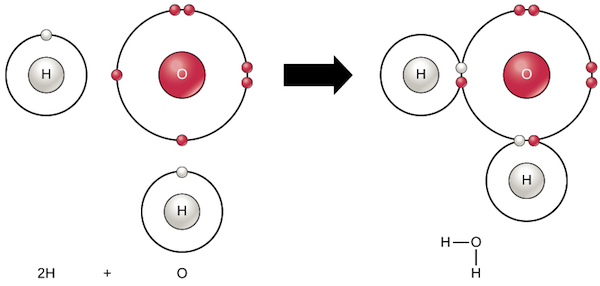
Covalent bonds occur where atoms share their valence electrons. Diagram of water molecules attached by hydrogen bonds (HKontro )Ĭovalent, ionic and hydrogen bonds are the most common bonds found in biological macromolecules. There are different types of bonds which can form depending on the charges of atoms and their electronegativities. Bonds are chemical connections that form between atoms to form larger molecules.
